🗺️ Anatomy of the urinary tract
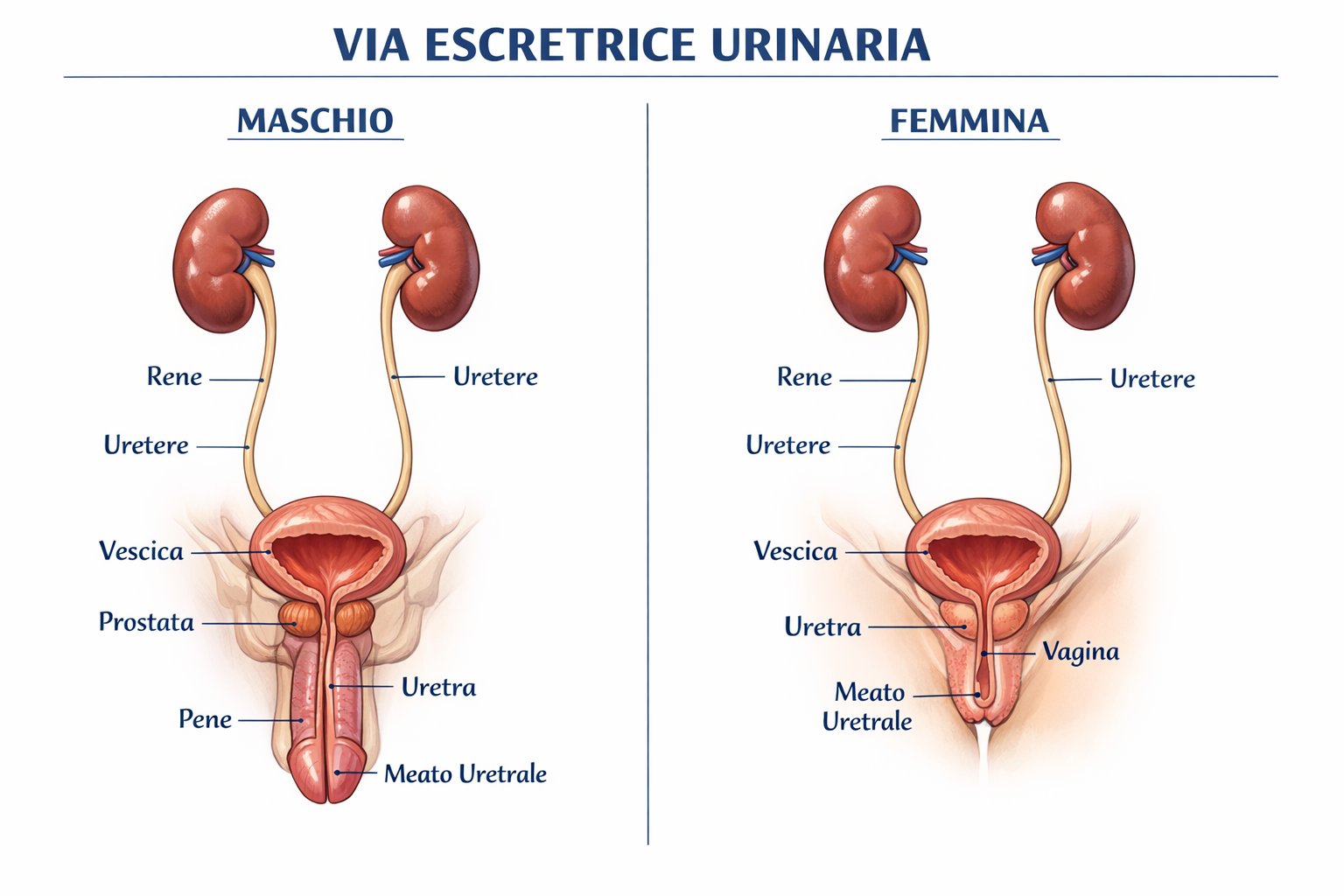
The upper urinary tract refers to the calyces, renal pelvis, and ureter down to the ureterovesical junction. By contrast, the lower urinary tract consists of the bladder and urethra.
The urothelial epithelium lines the entire urinary tract from the renal pelvis to the proximal urethra: it is the same cell type that generates tumours at all these sites — the basis for the concept of urothelial field disease.
📊 Epidemiology
UTUCs share the same risk factors as bladder carcinoma: cigarette smoking (principal), occupational exposure to aromatic amines, analgesics containing phenacetin (now withdrawn), and aristolochic acid (traditional Chinese herbal medicine — strongly associated with ureteric UTUC). The association with Lynch syndrome (mismatch repair gene deficiency — MLH1, MSH2) is significantly higher than in bladder carcinoma.
Urothelial field disease: patients with a prior UTUC carry a significant risk of developing metachronous bladder carcinoma (30–50%), whereas those with prior bladder cancer have a lower risk of developing UTUC (2–4%). This asymmetry is explained by the centrifugal flow of exfoliated urothelial cells.
🩺 Symptoms and diagnosis
Painless gross haematuria is the presenting symptom in 70–80% of UTUCs. In the presence of gross haematuria with a negative imaging workup for calculi, the differential diagnosis must include UTUC and mandates a multiphasic CT urogram with excretory phase.
Renal colic may be present when blood clots obstruct the renal pelvis or ureter, mimicking urolithiasis. This situation frequently causes diagnostic delay.
Reference standard. The excretory phase is essential for visualising the urothelial lumen. Sensitivity ~96% for lesions ≥5 mm.
Uretero-renal catheterisation with collection of urine from the ipsilateral pelvis. High specificity for high-grade tumours.
Direct visualisation of the lesion with a flexible ureteroscope. Forceps biopsy for histological typing and grading. Risk of seeding at the entry site (an argument in favour of early ureteral clipping during nephroureterectomy).
Always performed to exclude synchronous bladder lesions (field disease).
🔪 Radical nephroureterectomy — standard treatment
Radical nephroureterectomy (RNU) with excision of the bladder cuff is the standard treatment for non-metastatic UTUC not amenable to conservative management. It includes en-bloc removal of the kidney, perinephric fat, entire ureter down to the ureterovesical junction, and periostial bladder cuff.
- Standard in high-volume centres
- Reduced blood loss, shorter hospital stay
- Distal ureter management: intravesical or extravesical (stapling) approach
- Early ureteral clipping after vascular pedicle isolation (anti-seeding measure)
- Complete excision of ureter to the ostium — no ureteral stump should be left
- Locoregional lymphadenectomy for tumours ≥ pT2
- Single instillation of mitomycin C within 24 hours of RNU
- Reduces the risk of bladder recurrence by 40% (ODMIT-C trial)
- Now recommended by EAU Guidelines as standard practice
🎯 Conservative (kidney-sparing) treatment
In highly selected patients, a kidney-sparing approach aimed at preserving the ipsilateral kidney is feasible.
- Low histological grade (G1–G2)
- Small lesion (<1 cm, narrow implantation base)
- Location accessible by flexible ureteroscopy
- Functioning contralateral kidney
- High compliance with endoscopic surveillance
- Flexible ureteroscopy + laser (holmium/thulium)
- Ablation of the lesion under direct vision
- Biopsy of the residual base for verification
- Follow-up: ureteroscopy + cytology every 3 months for 2 years
🎓 Experience of the Padova Centre
The Urology group in Padova has accumulated one of the largest Italian series in conservative treatment of UTUC.
💊 Adjuvant chemotherapy — the POUT trial
The randomised POUT trial (Lancet 2020) demonstrated that adjuvant chemotherapy with gemcitabine + cisplatin (or carboplatin) for 4 cycles significantly reduces the risk of recurrence in patients with UTUC ≥ pT2 or N+ following radical nephroureterectomy.
The POUT regimen (gemcitabine + cisplatin or carboplatin according to GFR) is now recommended by the EAU Guidelines as the adjuvant standard for pT2–pT4 and/or N+ disease.
🌐 Metastatic disease
- Gemcitabine + cisplatin (cisplatin-eligible)
- Gemcitabine + carboplatin (cisplatin-ineligible)
- Dose-dense MVAC in fit patients
- Enfortumab vedotin (anti-Nectin-4 ADC) + pembrolizumab
- Efficacy demonstrated independent of renal function — crucially relevant in this post-RNU population
- New first-line standard for eligible metastatic UTUC
- Superior OS and PFS vs gemcitabine/platinum
The renal function-independent efficacy of EV + pembrolizumab is a particularly relevant feature in UTUC, where nephroureterectomy has already reduced renal reserve.